Product Center
-
● Automatic inspection machine for particulate matter in ampoules
-
● Automatic inspection machine for oral liquid foreign objects
-
● Automatic inspection machine for lyophilized preparations in vials
-
● Automatic foreign object inspection machine for plastic bottle intravenous infusions
-
● Electronic microporous leak detector for ampoule injections
-
● Fully automatic electronic microporous leak detector for infusion
-
● Automatic foreign object inspection machine for glass bottle intravenous infusions
-
● In-line plastic ampoule electronic micro-hole leak detector
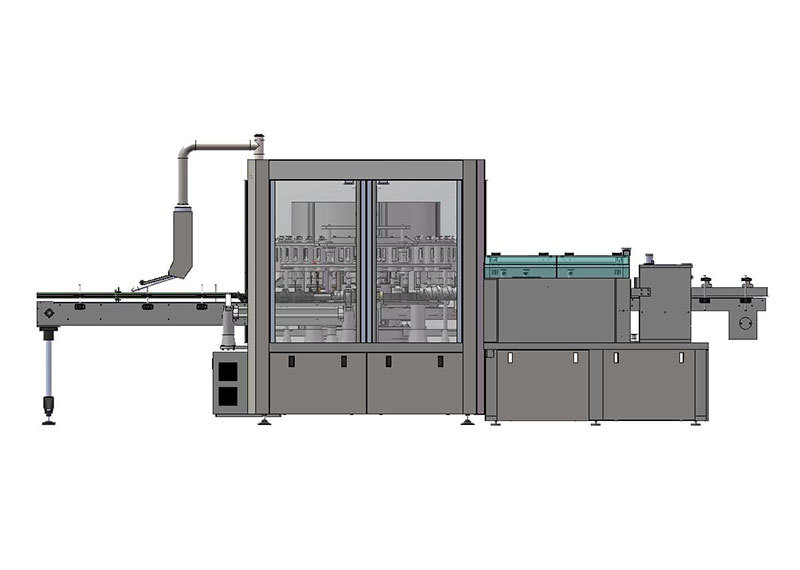
JSSZ/300 Plastic Bottle Large-Volume Infusion Automatic Foreign Object Inspection Machine
Mainly engaged in the research and development of ceramic new material valves, with metal valves (gate valves, globe valves, ball valves, butterfly valves) as a secondary business. Ceramic valves have been successfully applied in many new energy battery material plants, thermal power plants, nuclear power plants, chemical plants, steel plants, coal chemical industry, polysilicon and other industries, and have been listed as special-purpose products, contributing to energy conservation and environmental protection for enterprises and society.
Category
Automatic foreign object inspection machine for plastic bottle intravenous infusions

Details
Main Use
Mainly used for automatic foreign object inspection of plastic bottles for large-volume parenteral solutions produced in pharmaceutical factories. Inspection items include glass fragments, hair, white patches, white spots, and other foreign objects, as well as liquid level detection. Detection range: 50ml-250ml plastic and glass bottles for large-volume parenteral solutions.
Technical Parameters
Detection Speed: 0-300 bottles/minute
Applicable Specifications: 50-250ml large-volume parenteral solution plastic or glass bottles
Main Features
- Compact overall structure, simple design, easy maintenance, strong versatility
- The detection mechanism is located above the table, meeting GMP requirements
- Detection accuracy is adjustable to meet the production requirements of pharmaceutical factories
- Detection data can be retrieved, viewed, and stored, and has a printing interface. Ensures traceability of process parameters and meets GMP requirements
- Main key components, such as cameras, light sources, servo motors, main motors, frequency converters, proximity switches, controllers, transmission bearings, sliding bearings, etc., use imported or well-known domestic brands
- It adopts the Knapp test procedure recognized by the European Pharmacopoeia and the US FDA, making the main functions and parameters of the detection verifiable.
Mainly engaged in the research and development of ceramic new material valves, with metal valves (gate valves, globe valves, ball valves, butterfly valves) as a secondary business. Ceramic valves have been successfully applied in many new energy battery material plants, thermal power plants, nuclear power plants, chemical plants, steel plants, coal chemical industry, polysilicon and other industries, and have been listed as special-purpose products, contributing to energy conservation and environmental protection for enterprises and society.
Main Use
Mainly used for automatic foreign object inspection of plastic bottles for large-volume parenteral solutions produced in pharmaceutical factories. Inspection items include glass fragments, hair, white patches, white spots, and other foreign objects, as well as liquid level detection. Detection range: 50ml-250ml plastic and glass bottles for large-volume parenteral solutions.
Technical Parameters
Detection Speed: 0-300 bottles/minute
Applicable Specifications: 50-250ml large-volume parenteral solution plastic or glass bottles
Main Features
- Compact overall structure, simple design, easy maintenance, strong versatility
- The detection mechanism is located above the table, meeting GMP requirements
- Detection accuracy is adjustable to meet the production requirements of pharmaceutical factories
- Detection data can be retrieved, viewed, and stored, and has a printing interface. Ensures traceability of process parameters and meets GMP requirements
- Main key components, such as cameras, light sources, servo motors, main motors, frequency converters, proximity switches, controllers, transmission bearings, sliding bearings, etc., use imported or well-known domestic brands
- It adopts the Knapp test procedure recognized by the European Pharmacopoeia and the US FDA, making the main functions and parameters of the detection verifiable.
Related Products
Inquiry